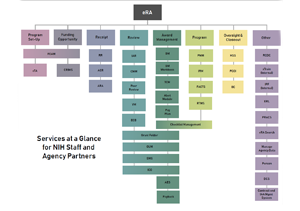
eRA provides a range of modules used by NIH and partner agency staff to help manage the receipt, processing, review, award, monitoring and reporting of research and non-research grants.
Program Set-Up
Modules that provide the ability for agencies to create and manage funding opportunities announcements online; and provide the ability for future year budgeting
Funding Opportunity
Modules that provide the ability for agencies to create and manage funding opportunities announcements online; and provide the ability for agency staff to electronically search for and request an activity code, the three-character code used to identify funding mechanisms (F32, R01, etc.)
Receipt
Modules that allow for receipt, triaging and assigning of grant applications; provide the ability to electronically track information received in advance of a grant application submission and link it to the application after the application is received; and provide the capability to electronically initiate, route and approve changes in application assignments.
Review
Modules that support the initial and secondary level review of grant applications; allow program staff to follow the application discussion in real time; allow reviewers to participate in virtual review meetings; and allow for the formation and administration of review committees.
Award Management
Modules that provide the ability for grantor agencies to perform the administrative review before issuing grant awards; calculate and check the appropriate amounts for all aspects of a grant award; define the terms and conditions that can be applied to the grants they administer; and request holds on grant records that they do not want archived.
Program
Modules that allow program staff to manage portfolios of grants and the end-to-end grant lifecycle; provide the ability to assign applications within an individual institute to program staff following the referral of the application to different institutes; allow an agency to accurately track and report their investments in research involving collaborations in foreign countries.
Oversight & Closeout
Modules that allow agencies to manage reports they receive from grantees on human subjects and clinical trials; reports they receive from institutions on any financial conflicts of interest involving their principal investigators; and reports required from grantees to close out a grant.
Other
Modules that do not fall into any of the above categories and include a module that allows NIH to categorize its funding in medical research each fiscal year for reporting to the public; a module that allows eRA’s partner agencies to manage certain data they own in the IMPAC II database; and more.




 eRA Intranet
eRA Intranet