Grantees — Submit Progress Reports
All progress reports for NIH grants must be submitted electronically using the Research Performance Progress Report (RPPR) module in eRA Commons (See OER’s RPPR webpage for details). Progress reports document the grantee recipient’s accomplishments and compliance with terms of award.
There are three types of RPPRs:
- Annual RPPR – Used to describe a grant’s scientific progress, identify significant changes, report on personnel, and describe plans for the subsequent budget period or year.
- Interim RPPR – Used when submitting a renewal (Type 2) application. If the Type 2 is not funded, the Interim RPPR will serve as the Final RPPR for the project. If the Type 2 is funded, the Interim RPPR will serve as the annual RPPR for the final year of the previous competitive segment. The data elements collected on the Interim RPPR are the same as for the Final RPPR, including project outcomes.
- Final RPPR – Used as part of the grant closeout process to submit project outcomes in addition to the information submitted on the annual RPPR, except budget and plans for the upcoming year.
Basic Tasks (step-by-step instructions)
- For Program Directors/Principal Investigators to initiate an RPPR
- For Signing Officials to submit an RPPR in eRA Commons
- For Signing Officials to delegate submission of an RPPR
- Submitting Your Interim Research Performance Progress Report
- Submitting Your Final Research Performance Progress Report
Main Screenshots
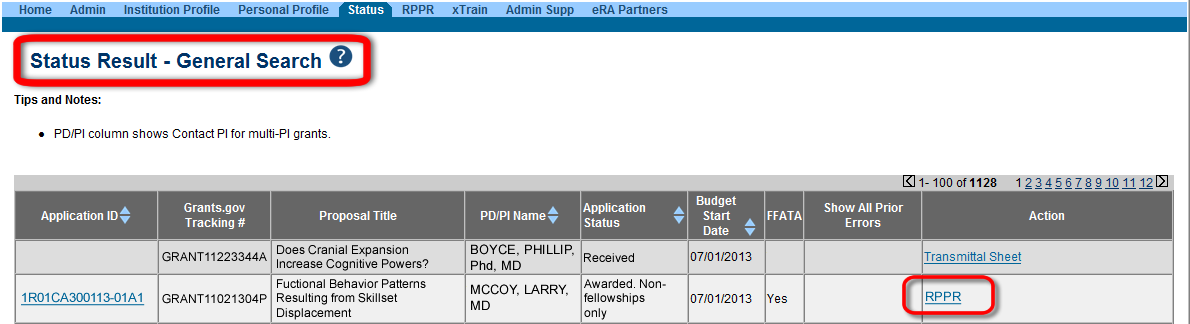
Figure 1: Accessing the annual RPPR through the Status screen
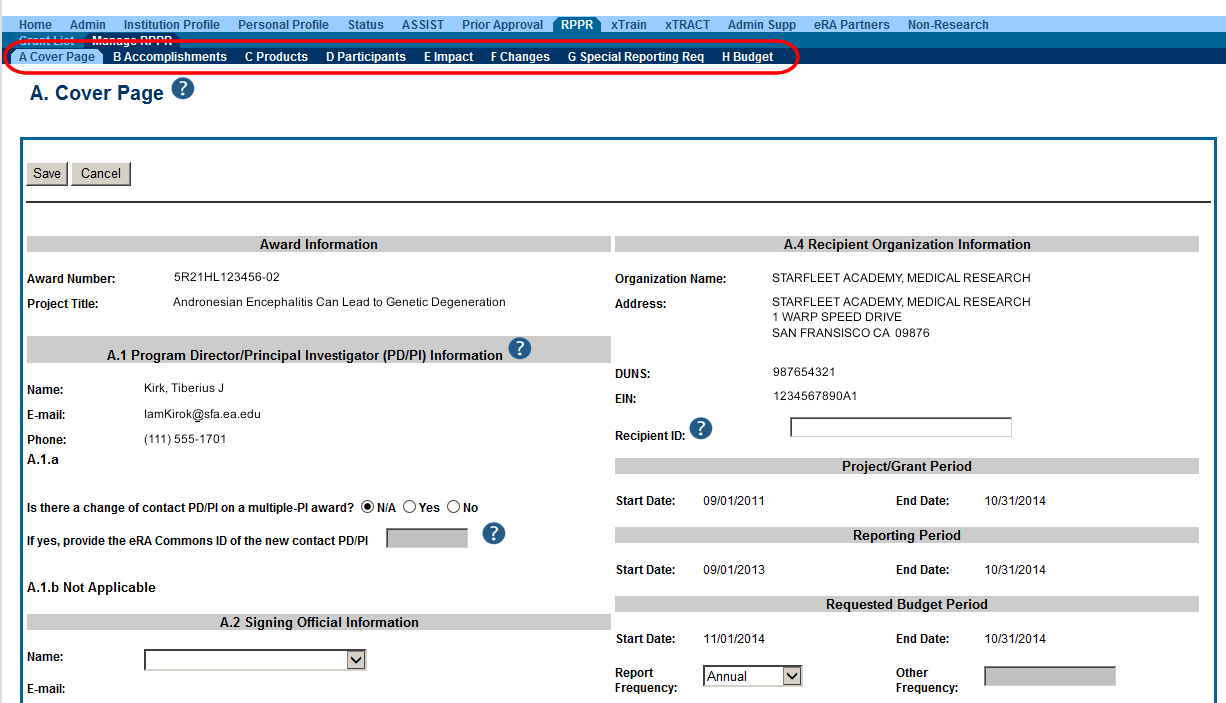
Figure 2: The Annual RPPR form and navigation tabs
Resources
- NIH's Implementation of the RPPR in eRA: An Overview
- RPPR Webpage (OER)
- RPPR online help
- RPPR User Guide
- Grants Closeout FAQs
- RPPR FAQs
- RPPR Training (webinar, slide presentations)
- RPPR: Who Can Do What? (PDF - 76KB)
- eRA Commons Roles & Privileges At a Glance (PDF - 25 KB)
- Grants Closeout in eRA Commons - (Video - 6:52 Min) [needs updating]
Policy Links
- NIH Grants Policy Statement: RPPR
- NIH Grants Policy Statement: Final Progress Report
- NIH Implementation of Final RPPR for SBIR/STTR: NOT-OD-17-085 (July 10, 2017)
- NIH Implementation of the Interim RPPR: NOT-OD-17-037 (Jan. 19, 2017)
- NIH Implementation of Final RPPR: NOT-OD-17-022 (Nov. 23, 2016)




 eRA Intranet
eRA Intranet